Title III – Expansion of Productive Capacity and Supply
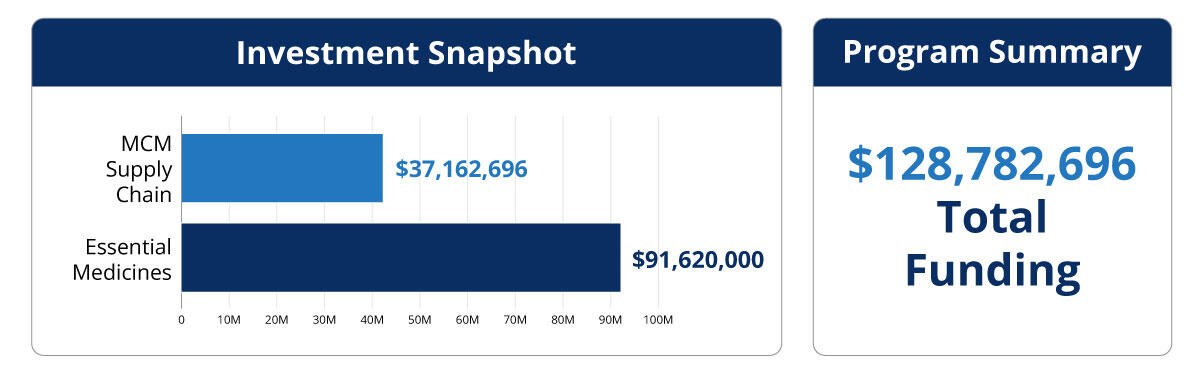
The U.S. Department of Health and Human Services’ (HHS) Defense Production Act (DPA) Title III Program is responsible for strengthening the National Defense by enhancing the resiliency, diversity, and security of supply chains for health resources deemed essential to prepare for, respond to, and recover from chemical, biological, radiological, and nuclear (CBRN) threats, and emerging infectious diseases (EIDs).
Programmatic Objectives
Expand domestic manufacturing capabilities: Enhance the ability of domestic manufacturers to produce and supply the Nation with health resources deemed by the President to be essential to the national defense.
Reduce supply chain vulnerabilities: Mitigate risks associated with global supply chain disruptions.
Promote innovation: Encourage the rapid development and commercialization of new technologies through research and development (R&D) initiatives.
Ensure affordability and accessibility: Ensure that the health resources being produced and supplied to the Nation are affordable and widely available to all.
Executive Orders Supported by HHS DPA Title III Program
The HHS DPA Title III Program supports and oversees contracts that further the goals defined in the following executive orders:
DPA-Related Executive Orders
HHS will leverage Defense Production Act (DPA) Title III authorities to strategically expand critical production, commercialize R&D efforts, and scale emerging technologies to support domestic public health resource manufacturing capabilities.
Solicitations
Requests for Information (RFIs)
| Status | Title | Response Date | Link |
|---|---|---|---|
| Current | There are no open RFIs at this time. | — | — |
| Past | Public Health Industrial Base Capabilities and Capacities – Essential Medicines | Sept 26, 2025 | View RFI |
| Past | Public Health Industrial Base – Anti-Microbials | Jan 22, 2025 | View RFI |
| Past | Public Health Industrial Base – Essential Medicines & MCMs | Apr 5, 2024 | View RFI |
Requests for Proposals (RFPs)
| Status | Title | Response Date | Link |
|---|---|---|---|
| Current | Request for Project Proposals: Production of Drug Substances and Drug Products at Commercial Scale – Anti-Microbials and Large Volume Parenterals | February 6, 2026 | March 9, 2026 |
| Past | RPP-24-02-KSM-API: Drug Substances & Products at Commercial Scale | Mar 20, 2024 | BioMap Consortium |
| Past | RPP-24-03-Sterilization: Capacity for Vaccines & Therapeutics | Mar 28, 20245 | BioMap Consortium |
Essential Medicines Portfolio
API Innovation Center (APIIC)

KSM/API and FDF
Antheia, Inc.

KSM/API and FDF
Manus Bio, Inc.

KSM/API and FDF
Medical Sterilization Portfolio
Meissner Corporation

Medical Sterilization
Sterigenics

Medical Sterilization
Steri-Tek

Medical Sterilization
DPA-Related Executive Orders
E.O. 13603: National Defense Resources Preparedness
E.O. 14001: A Sustainable Public Health Supply Chain
E.O. 14017: America's Supply Chains

